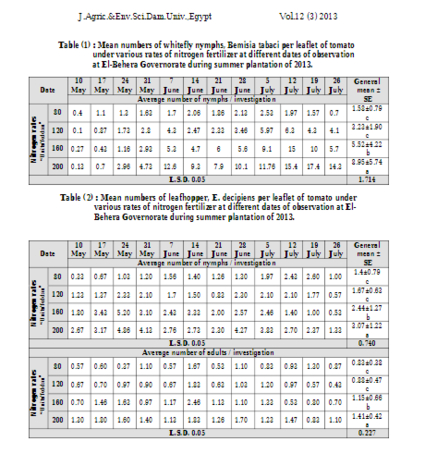
While transitioning to an anthroponic greenhouse, we need to calibrate the amount of nitrogen we're going to add to our system, taking into account that excessive fertilization will lead to the proliferation of sap-sucker insects. Indeed, many studies have shown the correlation between these two parameters: excessive nitrogen fertilization will cause luxuriant plant growth, making it more attractive to pests.
This implies that the added nitrogen should match the nitrogen uptake of the plant to avoid imbalances. We will only add the amount of nitrogen that the plants need: not more, not less. But how can we achieve this when this value changes based on the plant and its developmental stage?
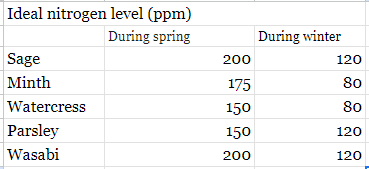
I've then checked the daily nitrogen uptake for the most common plants inside our greenhouse, including Mint, Sage, Wasabi, Watercress, and Parsley. The values differ from one plant to another but are all around 180 ppm during spring and around 100 ppm during winter.
Some studies show that aphid species thrive on plants fertilized with a solution of 250/300 ppm N. Therefore, the conclusion is quite immediate: we should keep the nitrogen concentration between 100-200 ppm, with higher levels during vegetative growth and the reproductive period and lower levels during the dormancy stage.
Next, I researched the composition of urine, but since it can greatly vary from one individual to another, directly testing the urine inside our collecting tank proved to be more effective.
The sample (5 mL) was diluted with 500 mL of water (at a ratio of 1:100), and tested for NH4+ content. The result was 1 ppm, indicating that the pure form contains 1000 ppm of ammonia (5 times more than our desired level). This means that, if we want to reach a 200 ppm concentration, we have to apply a dilution that's 5 times greater (0.5 mL of urine in 2500 mL of water).
Since our current system has 5000 L of water, we can calculate how much urine we need to add using the following proportion:
0.5 mL : 2500 mL = x : 5000000 mL
x = 1000 mL = 1 L
In conclusion, to reach a concentration of 200 ppm of nitrogen per day, we need to add on average 1 L of urine on a daily basis.
References: